Radiation therapy
Radiation Therapy for Cancer
What is radiation therapy?
Radiation therapy uses high-energy radiation to shrink tumors and kill cancer cells (1). X-rays, gamma rays, and charged particles are types of radiation used for cancer treatment.
The radiation may be delivered by a machine outside the body (external-beam radiation therapy), or it may come from radioactive material placed in the body near cancer cells (internal radiation therapy, also called brachytherapy).
Systemic radiation therapy uses radioactive substances, such as radioactive iodine, that travel in the blood to kill cancer cells.
About half of all cancer patients receive some type of radiation therapy sometime during the course of their treatment.
How does radiation therapy kill cancer cells?
Radiation therapy kills cancer cells by damaging their DNA (the molecules inside cells that carry genetic information and pass it from one generation to the next) (1). Radiation therapy can either damage DNA directly or create charged particles (free radicals) within the cells that can in turn damage the DNA.
Cancer cells whose DNA is damaged beyond repair stop dividing or die. When the damaged cells die, they are broken down and eliminated by the body’s natural processes.
Does radiation therapy kill only cancer cells?
No, radiation therapy can also damage normal cells, leading to side effects.
Doctors take potential damage to normal cells into account when planning a course of radiation therapy. The amount of radiation that normal tissue can safely receive is known for all parts of the body. Doctors use this information to help them decide where to aim radiation during treatment.
Why do patients receive radiation therapy?
Radiation therapy is sometimes given with curative intent (that is, with the hope that the treatment will cure a cancer, either by eliminating a tumor, preventing cancer recurrence, or both) (1). In such cases, radiation therapy may be used alone or in combination with surgery, chemotherapy, or both.
Radiation therapy may also be given with palliative intent. Palliative treatments are not intended to cure. Instead, they relieve symptoms and reduce the suffering caused by cancer.
Some examples of palliative radiation therapy are:
- Radiation given to the brain to shrink tumors formed from cancer cells that have spread to the brain from another part of the body (metastases).
- Radiation given to shrink a tumor that is pressing on the spine or growing within a bone, which can cause pain.
- Radiation given to shrink a tumor near the esophagus, which can interfere with a patient’s ability to eat and drink.
How is radiation therapy planned for an individual patient?
A radiation oncologist develops a patient’s treatment plan through a process called treatment planning, which begins with simulation.
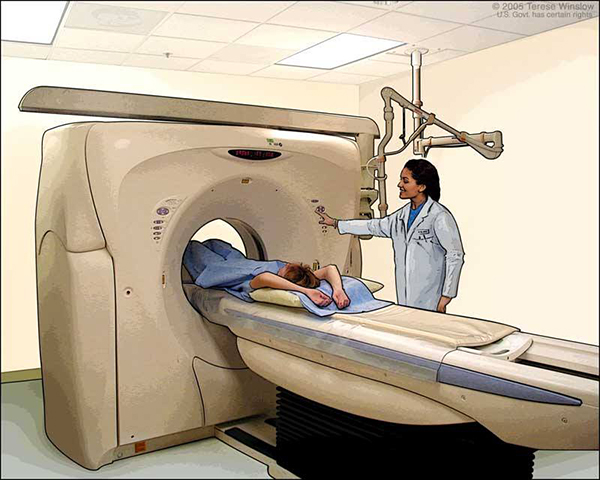
During simulation, detailed imaging scans show the location of a patient’s tumor and the normal areas around it. These scans are usually computed tomography (CT) scans, but they can also include magnetic resonance imaging (MRI), positron emission tomography(PET), and ultrasound scans.
CT scans are often used in treatment planning for radiation therapy. During CT scanning, pictures of the inside of the body are created by a computer linked to an x-ray machine.
During simulation and daily treatments, it is necessary to ensure that the patient will be in exactly the same position every day relative to the machine delivering the treatment or doing the imaging. Body molds, head masks, or other devices may be constructed for an individual patient to make it easier for a patient to stay still. Temporary skin marks and even tattoos are used to help with precise patient positioning.
Patients getting radiation to the head may need a mask. The mask helps keep the head from moving so that the patient is in the exact same position for each treatment.
After simulation, the radiation oncologist then determines the exact area that will be treated, the total radiation dose that will be delivered to the tumor, how much dose will be allowed for the normal tissues around the tumor, and the safest angles (paths) for radiation delivery.
The staff working with the radiation oncologist (including physicists and dosimetrists) use sophisticated computers to design the details of the exact radiation plan that will be used. After approving the plan, the radiation oncologist authorizes the start of treatment. On the first day of treatment, and usually at least weekly after that, many checks are made to ensure that the treatments are being delivered exactly the way they were planned.
Radiation doses for cancer treatment are measured in a unit called a gray (Gy), which is a measure of the amount of radiation energy absorbed by 1 kilogram of human tissue. Different doses of radiation are needed to kill different types of cancer cells.
Radiation can damage some types of normal tissue more easily than others. For example, the reproductive organs (testicles and ovaries) are more sensitive to radiation than bones. The radiation oncologist takes all of this information into account during treatment planning.
If an area of the body has previously been treated with radiation therapy, a patient may not be able to have radiation therapy to that area a second time, depending on how much radiation was given during the initial treatment. If one area of the body has already received the maximum safe lifetime dose of radiation, another area might still be treated with radiation therapy if the distance between the two areas is large enough.
The area selected for treatment usually includes the whole tumor plus a small amount of normal tissue surrounding the tumor. The normal tissue is treated for two main reasons:
- To take into account body movement from breathing and normal movement of the organs within the body, which can change the location of a tumor between treatments.
- To reduce the likelihood of tumor recurrence from cancer cells that have spread to the normal tissue next to the tumor (called microscopic local spread).
How is radiation therapy given to patients?
Radiation can come from a machine outside the body (external-beam radiation therapy) or from radioactive material placed in the body near cancer cells (internal radiation therapy, more commonly called brachytherapy). Systemic radiation therapy uses a radioactive substance, given by mouth or into a vein, that travels in the blood to tissues throughout the body.
The type of radiation therapy prescribed by a radiation oncologist depends on many factors, including:
- The type of cancer.
- The size of the cancer.
- The cancer’s location in the body.
- How close the cancer is to normal tissues that are sensitive to radiation.
- How far into the body the radiation needs to travel.
- The patient’s general health and medical history.
- Whether the patient will have other types of cancer treatment.
- Other factors, such as the patient’s age and other medical conditions.
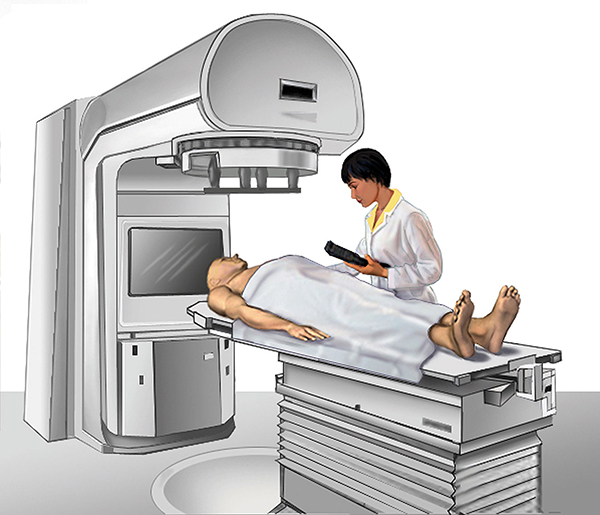
External-beam radiation therapy
External-beam radiation therapy is most often delivered in the form of photon beams (either x-rays or gamma rays) (1). A photon is the basic unit of light and other forms of electromagnetic radiation. It can be thought of as a bundle of energy. The amount of energy in a photon can vary. For example, the photons in gamma rays have the highest energy, followed by the photons in x-rays.
Many types of external-beam radiation therapy are delivered using a machine called a linear accelerator (also called a LINAC). A LINAC uses electricity to form a stream of fast-moving subatomic particles. This creates high-energy radiation that may be used to treat cancer.
Patients usually receive external-beam radiation therapy in daily treatment sessions over the course of several weeks. The number of treatment sessions depends on many factors, including the total radiation dose that will be given.
One of the most common types of external-beam radiation therapy is called 3-dimensional conformal radiation therapy (3D-CRT). 3D-CRT uses very sophisticated computer software and advanced treatment machines to deliver radiation to very precisely shaped target areas.
Many other methods of external-beam radiation therapy are currently being tested and used in cancer treatment. These methods include:
- Intensity-modulated radiation therapy (IMRT): IMRT uses hundreds of tiny radiation beam-shaping devices, called collimators, to deliver a single dose of radiation (2). The collimators can be stationary or can move during treatment, allowing the intensity of the radiation beams to change during treatment sessions. This kind of dose modulation allows different areas of a tumor or nearby tissues to receive different doses of radiation.Unlike other types of radiation therapy, IMRT is planned in reverse (called inverse treatment planning). In inverse treatment planning, the radiation oncologist chooses the radiation doses to different areas of the tumor and surrounding tissue, and then a high-powered computer program calculates the required number of beams and angles of the radiation treatment (3). In contrast, during traditional (forward) treatment planning, the radiation oncologist chooses the number and angles of the radiation beams in advance and computers calculate how much dose will be delivered from each of the planned beams.The goal of IMRT is to increase the radiation dose to the areas that need it and reduce radiation exposure to specific sensitive areas of surrounding normal tissue. Compared with 3D-CRT, IMRT can reduce the risk of some side effects, such as damage to the salivary glands (which can cause dry mouth, or xerostomia), when the head and neck are treated with radiation therapy (4). However, with IMRT, a larger volume of normal tissue overall is exposed to radiation. Whether IMRT leads to improved control of tumor growth and better survival compared with 3D-CRT is not yet known (4).
- Image-guided radiation therapy (IGRT): In IGRT, repeated imaging scans (CT, MRI, or PET) are performed during treatment. These imaging scans are processed by computers to identify changes in a tumor’s size and location due to treatment and to allow the position of the patient or the planned radiation dose to be adjusted during treatment as needed. Repeated imaging can increase the accuracy of radiation treatment and may allow reductions in the planned volume of tissue to be treated, thereby decreasing the total radiation dose to normal tissue (5).
- Tomotherapy: Tomotherapy is a type of image-guided IMRT. A tomotherapy machine is a hybrid between a CT imaging scanner and an external-beam radiation therapy machine (6). The part of the tomotherapy machine that delivers radiation for both imaging and treatment can rotate completely around the patient in the same manner as a normal CT scanner.Tomotherapy machines can capture CT images of the patient’s tumor immediately before treatment sessions, to allow for very precise tumor targeting and sparing of normal tissue.Like standard IMRT, tomotherapy may be better than 3D-CRT at sparing normal tissue from high radiation doses (7). However, clinical trials comparing 3D-CRT with tomotherapy have not been conducted.
- Stereotactic radiosurgery: Stereotactic radiosurgery (SRS) can deliver one or more high doses of radiation to a small tumor (5, 8). SRS uses extremely accurate image-guided tumor targeting and patient positioning. Therefore, a high dose of radiation can be given without excess damage to normal tissue.SRS can be used to treat only small tumors with well-defined edges. It is most commonly used in the treatment of brain or spinal tumors and brain metastases from other cancer types. For the treatment of some brain metastases, patients may receive radiation therapy to the entire brain (called whole-brain radiation therapy) in addition to SRS.SRS requires the use of a head frame or other device to immobilize the patient during treatment to ensure that the high dose of radiation is delivered accurately.
- Stereotactic body radiation therapy: Stereotactic body radiation therapy (SBRT) delivers radiation therapy in fewer sessions, using smaller radiation fields and higher doses than 3D-CRT in most cases. By definition, SBRT treats tumors that lie outside the brain and spinal cord. Because these tumors are more likely to move with the normal motion of the body, and therefore cannot be targeted as accurately as tumors within the brain or spine, SBRT is usually given in more than one dose (8). SBRT can be used to treat only small, isolated tumors, including cancers in the lung and liver (8).Many doctors refer to SBRT systems by their brand names, such as the CyberKnife®.
- Proton therapy: External-beam radiation therapy can be delivered by proton beams as well as the photon beams described above. Protons are a type of charged particle.Proton beams differ from photon beams mainly in the way they deposit energy in living tissue. Whereas photons deposit energy in small packets all along their path through tissue, protons deposit much of their energy at the end of their path (called the Bragg peak) and deposit less energy along the way.
- Other charged particle beams: Electron beams are used to irradiate superficial tumors, such as skin cancer or tumors near the surface of the body, but they cannot travel very far through tissue (1). Therefore, they cannot treat tumors deep within the body.
Patients can discuss these different methods of radiation therapy with their doctors to see if any is appropriate for their type of cancer and if it is available in their community or through a clinical trial.
Internal radiation therapy
Internal radiation therapy (brachytherapy) is radiation delivered from radiation sources (radioactive materials) placed inside or on the body (12). Several brachytherapy techniques are used in cancer treatment. Interstitial brachytherapy uses a radiation source placed within tumor tissue, such as within a prostate tumor. Intracavitary brachytherapy uses a source placed within a surgical cavity or a body cavity, such as the chest cavity, near a tumor. Episcleral brachytherapy, which is used to treat melanoma inside the eye, uses a source that is attached to the eye.
In brachytherapy, radioactive isotopes are sealed in tiny pellets or “seeds.” These seeds are placed in patients using delivery devices, such as needles, catheters, or some other type of carrier. As the isotopes decay naturally, they give off radiation that damages nearby cancer cells.
If left in place, after a few weeks or months, the isotopes decay completely and no longer give off radiation. The seeds will not cause harm if they are left in the body (see permanent brachytherapy, described below).
Brachytherapy may be able to deliver higher doses of radiation to some cancers than external-beam radiation therapy while causing less damage to normal tissue (1, 12).
Brachytherapy can be given as a low-dose-rate or a high-dose-rate treatment:
- In low-dose-rate treatment, cancer cells receive continuous low-dose radiation from the source over a period of several days (1, 12).
- In high-dose-rate treatment, a robotic machine attached to delivery tubes placed inside the body guides one or more radioactive sources into or near a tumor, and then removes the sources at the end of each treatment session. High-dose-rate treatment can be given in one or more treatment sessions.An example of a high-dose-rate treatment is the MammoSite® system, which is being studied to treat patients with breast cancer who have undergone breast-conserving surgery.
- For permament brachytherapy, the sources are surgically sealed within the body and left there, even after all of the radiation has been given off. The remaining material (in which the radioactive isotopes were sealed) does not cause any discomfort or harm to the patient. Permanent brachytherapy is a type of low-dose-rate brachytherapy.
- For temporary brachytherapy, tubes (catheters) or other carriers are used to deliver the radiation sources, and both the carriers and the radiation sources are removed after treatment. Temporary brachytherapy can be either low-dose-rate or high-dose-rate treatment.
Doctors can use brachytherapy alone or in addition to external-beam radiation therapy to provide a “boost” of radiation to a tumor while sparing surrounding normal tissue (12).
Systemic radiation therapy
In systemic radiation therapy, a patient swallows or receives an injection of a radioactive substance, such as radioactive iodine or a radioactive substance bound to a monoclonal antibody.
Radioactive iodine (131I) is a type of systemic radiation therapy commonly used to help treat some types of thyroid cancer. Thyroid cells naturally take up radioactive iodine.
For systemic radiation therapy for some other types of cancer, a monoclonal antibody helps target the radioactive substance to the right place. The antibody joined to the radioactive substance travels through the blood, locating and killing tumor cells. For example:
- The drug ibritumomab tiuxetan (Zevalin®) has been approved by the Food and Drug Administration (FDA) for the treatment of certain types of B-cell non-Hodgkin lymphoma (NHL). The antibody part of this drug recognizes and binds to a protein found on the surface of B lymphocytes.
- The combination drug regimen of tositumomab and iodine I 131 tositumomab(Bexxar®) has been approved for the treatment of certain types of NHL. In this regimen, nonradioactive tositumomab antibodies are given to patients first, followed by treatment with tositumomab antibodies that have 131I attached. Tositumomab recognizes and binds to the same protein on B lymphocytes as ibritumomab. The nonradioactive form of the antibody helps protect normal B lymphocytes from being damaged by radiation from 131I.
Many other systemic radiation therapy drugs are in clinical trials for different cancer types.
Some systemic radiation therapy drugs relieve pain from cancer that has spread to the bone (bone metastases). This is a type of palliative radiation therapy. The radioactive drugs samarium-153-lexidronam (Quadramet®) and strontium-89 chloride (Metastron®) are examples of radiopharmaceuticals used to treat pain from bone metastases (13).
Why are some types of radiation therapy given in many small doses?
Patients who receive most types of external-beam radiation therapy usually have to travel to the hospital or an outpatient facility up to 5 days a week for several weeks. One dose (a single fraction) of the total planned dose of radiation is given each day. Occasionally, two treatments a day are given.
Most types of external-beam radiation therapy are given in once-daily fractions. There are two main reasons for once-daily treatment:
- To minimize the damage to normal tissue.
- To increase the likelihood that cancer cells are exposed to radiation at the points in the cell cycle when they are most vulnerable to DNA damage (1, 14).
In recent decades, doctors have tested whether other fractionation schedules are helpful (1), including:
- Accelerated fractionation—treatment given in larger daily or weekly doses to reduce the number of weeks of treatment.
- Hyperfractionation—smaller doses of radiation given more than once a day.
- Hypofractionation—larger doses given once a day or less often to reduce the number of treatments.
Researchers hope that different types of treatment fractionation may either be more effective than traditional fractionation or be as effective but more convenient.
When will a patient get radiation therapy?
A patient may receive radiation therapy before, during, or after surgery. Some patients may receive radiation therapy alone, without surgery or other treatments. Some patients may receive radiation therapy and chemotherapy at the same time. The timing of radiation therapy depends on the type of cancer being treated and the goal of treatment (cure or palliation).
Radiation therapy given before surgery is called pre-operative or neoadjuvant radiation. Neoadjuvant radiation may be given to shrink a tumor so it can be removed by surgery and be less likely to return after surgery (1).
Radiation therapy given during surgery is called intraoperative radiation therapy (IORT). IORT can be external-beam radiation therapy (with photons or electrons) or brachytherapy. When radiation is given during surgery, nearby normal tissues can be physically shielded from radiation exposure (15). IORT is sometimes used when normal structures are too close to a tumor to allow the use of external-beam radiation therapy.
Radiation therapy given after surgery is called post-operative or adjuvant radiation therapy.
Radiation therapy given after some types of complicated surgery (especially in the abdomen or pelvis) may produce too many side effects; therefore, it may be safer if given before surgery in these cases (1).
The combination of chemotherapy and radiation therapy given at the same time is sometimes called chemoradiation or radiochemotherapy. For some types of cancer, the combination of chemotherapy and radiation therapy may kill more cancer cells (increasing the likelihood of a cure), but it can also cause more side effects (1, 14).
After cancer treatment, patients receive regular follow-up care from their oncologists to monitor their health and to check for possible cancer recurrence. Detailed information about follow-up care can be found at NCI's Follow-up Medical Care page.
Does radiation therapy make a patient radioactive?
External-beam radiation does not make a patient radioactive.
During temporary brachytherapy treatments, while the radioactive material is inside the body, the patient is radioactive; however, as soon as the material is removed, the patient is no longer radioactive. For temporary brachytherapy, the patient will usually stay in the hospital in a special room that shields other people from the radiation.
During permanent brachytherapy, the implanted material will be radioactive for several days, weeks, or months after the radiation source is put in place. During this time, the patient is radioactive. However, the amount of radiation reaching the surface of the skin is usually very low. Nonetheless, this radiation can be detected by radiation monitors and contact with pregnant woman and young children may be restricted for a few days or weeks.
Some types of systemic radiation therapy may temporarily make a patient’s bodily fluids (such as saliva, urine, sweat, or stool) emit a low level of radiation. Patients receiving systemic radiation therapy may need to limit their contact with other people during this time, and especially avoid contact with children younger than 18 and pregnant women.
A patient’s doctor or nurse will provide more information to family members and caretakers if any of these special precautions are needed. Over time (usually days or weeks), the radioactive material retained within the body will break down so that no radiation can be measured outside the patient’s body.
What are the potential side effects of radiation therapy?
Radiation therapy can cause both early (acute) and late (chronic) side effects. Acute side effects occur during treatment, and chronic side effects occur months or even years after treatment ends (1). The side effects that develop depend on the area of the body being treated, the dose given per day, the total dose given, the patient’s general medical condition, and other treatments given at the same time.
Acute radiation side effects are caused by damage to rapidly dividing normal cells in the area being treated. These effects include skin irritation or damage at regions exposed to the radiation beams. Examples include damage to the salivary glands or hair loss when the head or neck area is treated, or urinary problems when the lower abdomen is treated.
Most acute effects disappear after treatment ends, though some (like salivary glanddamage) can be permanent. The drug amifostine (Ethyol®) can help protect the salivary glands from radiation damage if it is given during treatment. Amifostine is the only drug approved by the FDA to protect normal tissues from radiation during treatment. This type of drug is called a radioprotector. Other potential radioprotectors are being tested in clinical trials.
Fatigue is a common side effect of radiation therapy regardless of which part of the body is treated. Nausea with or without vomiting is common when the abdomen is treated and occurs sometimes when the brain is treated. Medications are available to help prevent or treat nausea and vomiting during treatment.
Late side effects of radiation therapy may or may not occur. Depending on the area of the body treated, late side effects can include (1):
- Fibrosis (the replacement of normal tissue with scar tissue, leading to restricted movement of the affected area).
- Damage to the bowels, causing diarrhea and bleeding.
- Memory loss.
- Infertility (inability to have a child).
- Rarely, a second cancer caused by radiation exposure.
Second cancers that develop after radiation therapy depend on the part of the body that was treated (16). For example, girls treated with radiation to the chest for Hodgkin lymphoma have an increased risk of developing breast cancer later in life. In general, the lifetime risk of a second cancer is highest in people treated for cancer as children or adolescents (16).
Whether or not a patient experiences late side effects depends on other aspects of their cancer treatment in addition to radiation therapy, as well as their individual risk factors. Some chemotherapy drugs, genetic risk factors, and lifestyle factors (such as smoking) can also increase the risk of late side effects.
When suggesting radiation therapy as part of a patient’s cancer treatment, the radiation oncologist will carefully weigh the known risks of treatment against the potential benefits for each patient (including relief of symptoms, shrinking a tumor, or potential cure). The results of hundreds of clinical trials and doctors’ individual experiences help radiation oncologists decide which patients are likely to benefit from radiation therapy.
A more comprehensive discussion of acute and late side effects from radiation therapy, as well as ways to cope with these side effects, can be found in the NCI publications Radiation Therapy and You: Support for People With Cancer and the Radiation Therapy Side Effects Series.
What research is being done to improve radiation therapy?
Doctors and other scientists are conducting research studies called clinical trials to learn how to use radiation therapy to treat cancer more safely and effectively. Clinical trials allow researchers to examine the effectiveness of new treatments in comparison with standard ones, as well as to compare the side effects of the treatments.
Researchers are working on improving image-guided radiation so that it provides real-time imaging of the tumor target during treatment. Real-time imaging could help compensate for normal movement of the internal organs from breathing and for changes in tumor size during treatment.
Researchers are also studying radiosensitizers and radioprotectors, chemicals that modify a cell's response to radiation:
- Radiosensitizers are drugs that make cancer cells more sensitive to the effects of radiation therapy. Several agents are under study as radiosensitizers. In addition, some anticancer drugs, such as 5-fluorouracil and cisplatin, make cancer cells more sensitive to radiation therapy.
- Radioprotectors (also called radioprotectants) are drugs that protect normal cells from damage caused by radiation therapy. These drugs promote the repair of normal cells exposed to radiation. Many agents are currently being studied as potential radioprotectors.
The use of carbon ion beams in radiation therapy is being investigated by researchers, but, at this time, the use of these beams remains experimental. Carbon ion beams are available at only a few medical centers around the world. They are not currently available in the United States. Researchers hope that carbon ion beams may be effective in treating some tumors that are resistant to traditional radiation therapy.
People with cancer who are interested in taking part in a clinical trial should talk with their doctor. A comprehensive list of current clinical trials is available on NCI’s website.
NCI's Cancer Information Service (CIS) can also provide information about clinical trials and help with clinical trial searches. Call the CIS at 1–800–4–CANCER (1–800–422–6237).


Comments
Post a Comment